Vulnerable 5-11 year-olds in Northern Ireland offered coronavirus vaccination
and live on Freeview channel 276
Parents of children aged 5-11 who are considered at higher risk of infection received letters from the Department of Health over the weekend, inviting them to bring their children forward for the vaccines.
This includes children with severe neurodisabilities, children with immunosupression, profound and multiple or severe learning disabilities, children with Down’s syndrome, and other long-term serious conditions.
Advertisement
Hide AdAdvertisement
Hide AdThe letter from the Department of Health also recommends vaccination for 5-11 year-olds living with “people who have a weakened immune system”.
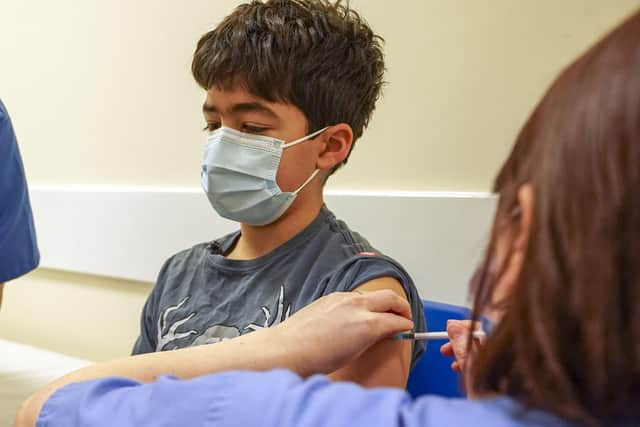

“This is to reduce the risk of them passing on the infection to their family members,” the letter states.
The Pfizer vaccine is being offered. The dosage is 10 micrograms — a third of the amount used for adults.
“Your child needs two injections of the vaccine, usually eight weeks apart,” the letter states. “The vaccine has been tested to make sure it is as safe as possible.”
Advertisement
Hide AdAdvertisement
Hide AdAcross the UK, around 500,000 children in the age bracket, who are either most at risk of Covid-19 or live with someone vulnerable, are now able to get their vaccinations.
The expansion of the programme comes two weeks after the UK vaccine roll-out was further expanded so all 16 and 17-year-olds could have a booster.
Dr Jillian Johnston, Joint Interim Assistant Director in Health Protection at the PHA, said in a statement: “There are approximately 25,000 children aged 5 to 11 year olds in Northern Ireland eligible for the COVID-19 vaccine and we are urging the parents of these children to take up the offer when invited.
“A new specially designed paediatric formulation of the Pfizer BioNTech COVID-19 vaccine has been approved for this age group and it’s given at a lower dose compared to that used in individuals aged 12 and above (10 micrograms compared with 30 micrograms). Like all children’s vaccines this approval was given following a robust review of data showing that it is safe and effective to use in this cohort.”
Advertisement
Hide AdAdvertisement
Hide AdGPs are also writing to the parents of eligible children. The Public Health Agency is asking parents of eligible children to look out for the letter, and speak to their GP if it hasn’t been received by “early February”.
Eligible children will either receive their vaccine via dedicated Trust vaccine clinics or in school for those children who attend special schools, the Public Health Agency has said.